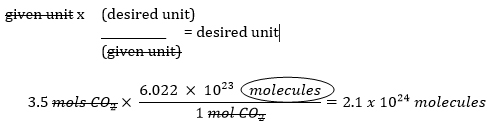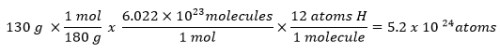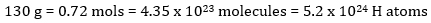Moles represent or stand in for other quantities. One such quantity is mass. The atomic mass (for for an element) and the formula weight (for a compound) are equal to a mole.
Related Byte: Moles to grams conversions
But moles can also represent the number of:
- Atoms (for elements)
- Molecules (for compounds)
NOTE: atoms and molecules are different and moles will therefore represent different quantities depending on if you have a compound or element.
- One mole is equivalent to “x” value of atoms of an element
- One mole is equivalent to “x” value of molecules of a compound
What is “x?” The number of atoms or molecules equal to one mole is called Avogadro’s Number, and its value is 6.022 x 1023.
- 1 mole of an ELEMENT = 6.022 x 1023 ATOMS
- 1 mole of COMPOUND = 6.022 x 1023 MOLECULES
Do these look familiar? They should—they are conversion factors!
You can write them as follows:
Starting with Moles
Use the proper Avogadro’s number conversion factor and dimensional analysis to solve in one simple step.
Steps:
Example: How many molecules are in 3.5 mols of CO2?
Solve using dimensional analysis:
Recall the set up of dimensional analysis such that given units are positioned to cancel out!
Related Byte: Dimensional Analysis
Starting with Grams
So what if you have to find the number of molecules beginning with a mass rather than moles? Simple, just one more step.
There are __________ molecules in 130 grams of C6H12O6?
Calculating Atoms from Molecules for a Compound
You can find the number of atoms of a particular element or all elements in a molecule of a compound. For example, in CO3, there is 1 atom of C and 3 atoms of O or a total of 4 atoms altogether.
In C6H12O6 , there are 6 atoms of C, 12 atoms of H, and 6 atoms of O or a total of 24 atoms in the entire molecule, etc.
These are also conversion factors. Let’s try an example, using conversion factors and dimensional analysis.
Steps:
How many hydrogen atoms are there in 130 grams of C6H12O6?
All at once:
Step by step: