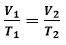Definition: Charles’ Law describes the relationship between Temperature (T) and Volume (V) while the number of moles (n) and pressure (P) remain constant.
Relation: T and V are directly proportional so as temperature increases, volume increases—and vise versa
T α V
Equation:
Example:
Up, Up, and Away! A hot-air balloon uses Charles’ Law to help it fly. As the gas inside the balloon is heated (↑ T), the balloon inflates (↑V). The warm air then rises, and so does the balloon.
http://commons.wikimedia.org/wiki/File:Flames_from_burner_in_hot_air_balloon.JPG
Problem: A sample of carbon monoxide gas occupies 3.20 L at 125°C. At what temperature will the gas occupy a volume of 1.54 L if the pressure remains constant?
GIVEN:
V1=3.2L
T1= 125°C → K Conversion Required → 125°C + 273 = 398K
T2= ?
V2=1.54L