Identify which element’s charge increased. That atom has been oxidized. The atom which has been reduced will see a decrease in its charge.
- Oxidized element: charge increases; also called the reducing agent
- Reduced element: charge decreases; also called the oxidizing agent
- The element which was oxidized will act as the reducing agent for the other element.
- The element which was reduced will act as the oxidizing agent for the other element.
Let’s try an example:
| Element | Oxidization Number BEFORE | Oxidization Number AFTER |
| Zn | 0 | +2 |
| Cu | +2 | 0 |
Q: Which element was oxidized and why?
A: Zinc because its charge increased from 0 to +2.
Q: Which element was reduced and why?
A: Copper because its charge decreased from +2 to 0.
Q: What is the oxidizing agent?
A: Copper was reduced, making it an oxidizing agent (it oxidized Zn).
Q: What is the reducing agent?
A: Zinc was oxidized, making it a reducing agent (it reduced Cu).
Q: How many electrons were exchanged?
A: Two electrons (because the difference between 0 and +2 is 2).
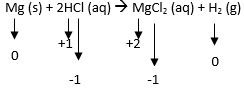
Example:
| Element | Oxidization Number BEFORE | Oxidization Number AFTER |
| Mg | 0 | +2 |
| H | +1 | 0 |
| Cl | -1 | -1 |
Related Byte: Redox Reaction Basics, Activity Series
Q: Which element was oxidized and why?
A: Magnesium because its charge increased from 0 to +2.
Q: Which element was reduced and why?
A: Hydrogen because its charge decreased from +1 to 0.
Q: Which element is the spectator ion and why?
A: Chlorine because the charge did not change. It was -1 before and after the reaction.
Q: What is the oxidizing agent?
A: H was reduced, making it an oxidizing agent (it oxidized Mg).
Q: What is the reducing agent?
A: Mg was oxidized, making it a reducing agent (it reduced H).
Q: How many electrons were exchanged?
A: Two electrons (because Mg lost 2 electrons and each of the 2 H gained one electron).