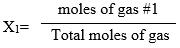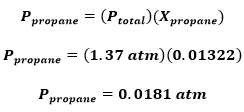Definition: Dalton’s Law of Partial Pressures states that the total pressure of a gas mixture is equal to the sum of all individual or partial pressures of its component gases.
Ptotal = P1 + P2 + P3
Partial pressures are proportional to each gas’ mole fraction (how much of the gas is present in the mixture).
To find Mole Fraction:
To find the Partial Pressure for each gas in mixture:
P1= (Ptotal) (X1) ….and so on for each gas in the mixture
To find Total Pressure: Ptotal = P1 + P2 + P3
Problem: A sample of natural gas contains 8.24 moles of CH4, 0.421 moles of C2H6, and 0.116 moles of C3H8. If the total pressure of the gases is 1.37 atm, what is the partial pressure of propane (C3H8)?
GIVEN:
Moles CH4 = 8.24
Moles C2H6 = 0.421
Moles C3H8 = 0.116
PTotal = 1.37 atm
Part I: Find total number of moles
8.24 + 0.421 + 0.116 = 8.777 mols
Part II: Find the mole fraction for proprane (C3H8)
Part III: Find the partial pressure of proprane (C3H8)