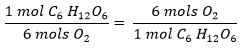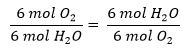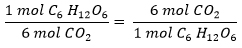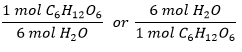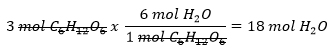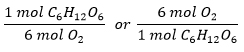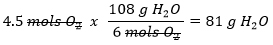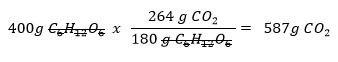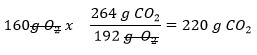Think of chemical reactions like recipes. What does this mean? Let’s explore…
Consider a rice mix which requires you to add 1- 2/3 cups of water and 1 tablespoon of butter to one box of the mixture. These ingredients are all of your reactants.
In return, the directions state, it should produce a 2 cups of rice. Depending on how much rice you want to make (2 cups, 4 cups, 6 cups, etc), you should vary the amount of ingredients.
Now using a chemical reaction…
C6H12O6 + 6O2 → 6CO2 + 6H2O
The coefficients represents the # MOLES. So this equation tells us the following:
- 1 mol C6H12O6 requires 6 mols of O2 to react
- For each 1 mol C6H12O6 and 6 mols of O2, 6 mols CO2 and 6 mols of H2O are produced
- For each 6 mols of CO2 that are produced, 6 mols of H2O are also made
This is a conversion factor between glucose and oxygen!
And for oxygen and water?
And for glucose and carbon dioxide?
THESE (AND MORE!) ARE ALL CONVERSION FACTORS AND CAN BE USED TO DETERMINE REACTION YIELD.
The purpose of these balanced equations is to tell you the proper proportions of reactants and products. You should then determine the correct and appropriate conversion factor for use in the given problem to solve it using dimensional analysis.
Calculations Using Moles Only:
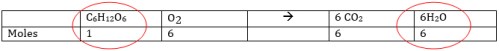
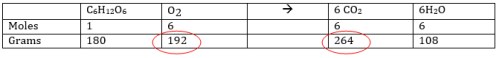
Fill in the following table to extract the # moles of each reactant and product in the equation. These will be the mole to mole ratios you will use in conversion factor to solve problems.
C6H12O6 + 6O2 → 6CO2 + 6H2O
Now complete the following problems using the information in the table:
How many moles of H2O can be produced from 3 moles of C6H12O6?
- This is reactant: product relationship
- Relation is between H2O and C6H12O6
- Conversion factors are:
- Set up using the given amount: 3 mols C6H12¬O6 and solve using dimensional analysis
C6H12O6 + 6O2 → 6CO2 + 6H2O
How many moles of C6H12O6 are required to react with 8 moles of O2?
- This is a reactant: reactant relationship
- Relation is between C6H12O6 and O2
- Conversion factors are:
- Solve

Calculations Using Grams and Moles:
If you are asked to calculate the yield in mass, you will need to convert the mole values in the table to reflect the mass equivalent to the # of moles! These will be the mole to grams ratios from which you can select to form conversion factor to solve problems.
C6H12O6 + 6O2 → 6CO2 + 6H2O
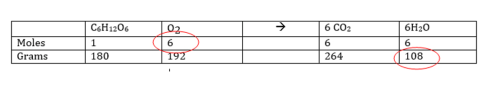
Now complete the following problem using the information in the table:
How many grams of H2O can be produced from 4.5 mols of O2?
Calculations Using Grams Only:
Derive the same table as before, but this time, values of interest will be used for gram to gram ratios from which you can select to form conversion factor to solve problems.
C6H12O6 + 6O2 → 6CO2 + 6H2O
 How many grams of CO2 can be produced from 400g of C6H12O6?
How many grams of CO2 can be produced from 400g of C6H12O6?
Calculations with More Than One Reactant:
What if you were given more that 1 reactant amount? You would have to calculate the yield using each amount and then report the smallest yield as your result. Why? Because once you run out of one of your reactants, the reaction shuts down and no further product is made.
The reactant that limits the amount of product made and is itself consumed in its entirety is called the limiting reagent.
So let’s try the same problem as above which gave the grams of C6H12O6 only and add consider that we must also factor in the grams of O2 present during the reaction. How will that change the amount of CO2 formed?
We must solve the problem twice:
- once using amount of C6H12O6
- once using amount of O2
How many grams of CO2 can be produced from 400g of C6H12O6 and 160g of O2?
- We already know from the previous example that from the 400 g of C6H12O6, 587 g of CO2 were formed
- Now perform the same calculation to determine how much CO2 is formed from 160 g of O2
C6H12O6 + 6O2 → 6 CO2 + 6H2O
- So from 160g of O2, 220 grams of CO2 can be produced versus the 587g produced from the 400g of C6H12O6. Which value is true? The smaller one!
- Why? Because you must report the amount yielded from the limiting reagent which is O2. It produced the lesser amount: 220 g.
- Once the 220g of CO2 is produced, the reaction runs out of O2 and therefore stops! So no more product is made, regardless of how much C6H12O6 still remains in excess.