Bonding and Polarity 101
Types of Bonds:
Ionic Bonds
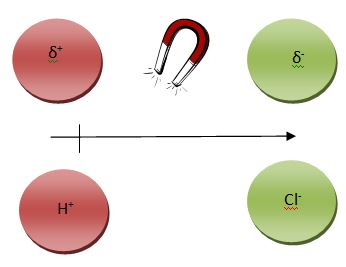
Ion-Ion Interactions that occur within a compound between a metal (cation) and a nonmetal (anion) an ionic bond. This occurs when there is a net loss and gain of electrons from the cation to the anion. Hence, electrons are transferred.
The level of attraction between the two ions is great, making ionic bonds the strongest bonds. NaCl is a posterchild example of such a bond, a salt composed of a metal and a nonmetal. This is a match made in chemistry heaven.
Covalent Bonds
Partially charged atoms are called dipoles and may be either be positive dipoles δ+or negative dipoles δ–. They can also be noted by a single-headed arrow, whose head points towards the more – ion, the δ-, and a + tail which points towards the δ+. The term “dipole” simply means two (di) poles (opposite poles, a positive and a negative).
This type of bonds occurs between nonmetals.
Dipoles of two nonmetals within a compound are called covalent bonds, which while strong, are not as strong ionic bonds.. This occurs when there is not net loss or gain of electrons, or transfer, but rather, the electrons are shared between the two atoms.
There are two types of covalent bonds:
- Polar covalent
- Nonpolar covalent
Nonpolar Covalent Bonds:
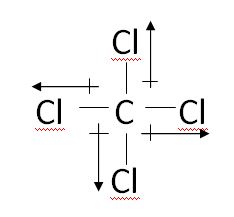
- Are symmetrical, which means that the central atom is BOTH surrounded by all the same type of atoms and possesses no lone pairs of electrons
- Contain no dipole moments because all of their dipoles cancel one another out. This means that the electronegativity and partial charges are evenly distributed throughout the atom
Carbon tetrachloride is an example of a nonpolar compound because the dipoles are balanced. Notice that all dipoles (up/down and left/right) cancel one another out. Also, the central atom, carbon, is surrounded by all hydrogen atoms and contains NO lone pairs of electrons.
Polar Covalent Bonds:
- Are asymmetrical, which means that the central atom may not be surrounded by all the same atom or that it may possess lone pairs of electrons
- Contains a dipole moment, which means that its dipoles are not symmetrical and do not cancel one another out
- Do not have the electronegativities and partial charges evenly distributed
Polarity can also be determined using dipoles (and dipole moments) and electronegativity differences.
Summary:
Ionic Bonds
- Occur between metals and nonmetals
- Strongest bond
- Involves the exchange (loss and gain) of electrons
- Occur between fully charged ions
Covalent Bonds
- Occur between nonmetals
- Weaker than ionic bond
- Involves the sharing of electrons (equally in nonpolar and unequally in polar)
- Occur between partial charges (dipoles)