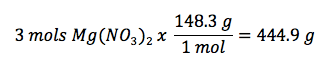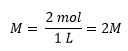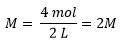Solutions are made at varying concentrations. We have many units to signify concentration, the amount of solute to solution ratio. Perhaps the most common concentration unit in chemistry is molarity, abbreviated (M).
Molarity is the defined as the number of moles of solute per liter of total solution.
For example, a 2M solution of NaCl means that 2 moles of NaCl (equivalent to 117 grams) is dissolved in enough water to make 1 liter of solution.
NOTE: The solute is NOT added to 1 L of water—that would result in a volume of more than 1L because the 117 grams of NaCl takes up some volume. So we add less than 1L of water, technically.
If you wanted to make 2L of this solution, you would have to use twice as much solute: 4 mols (234 grams) and add enough water to make 2L of solution.
You could also make 0.5 L or 500 mL, in which case you would take half as much solute: 0.5 mols (29.25 grams) and add enough water to make 500 mL of solution.
All of these scenarios would make the same molarity because the ratios would be equal.
You will have to rely on gram to mole conversions to calculate molarity properly. Also, don’t forget to use liters as your unit of volume—you may have to convert from mL to L in some problems!
Related Byte: mL to L conversion and gram to mole conversions
Example: What is the molarity of a solution containing 345 grams of CaCl2 dissolved enough water to make 750 mL?
Given:
- Mass = 345 grams CaCl2 → convert to moles
- Volume = 750 mL → convert to Liters
Conversions:
750 mL = 0.750 L
Solve:

Example: How many grams of Mg(NO3)2 should be used to form 2 liters of a 1.5M solution?
Given:
- Volume = 2L
- Molarity = 1.5M
Solve:
Convert: