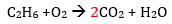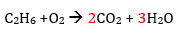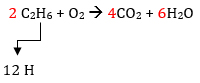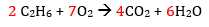The Law of Conservation of Mass states that matter can neither be created nor destroyed. Therefore, the # of atoms of each element must be equal on both sides of the equation.
The left half of the equation is called the “reactants” and the right side are the “products.” So we have 1 A on each side of equation and 2 B atoms on each side.
Coefficients are used in front of each formula to change the # of atoms. A coefficient is multiplied or distributed to all atoms that follow it.
Here are some other examples:
Guidelines to Balance Chemical Equations:
- The # of all atoms must be the same on both sides of the arrow.
- Begin with all NON oxygen and hydrogen atoms…those will almost always work themselves out. Also begin with the element which appears in only ONE reactant or product (this is usually the NON oxygen and hydrogen element(s)
- Use only COEFFICIENTS to balance the equation. DO NOT CHANGE SUBSCRIPTS.
- It is easier to balance polyatomic ions as units rather than as individual atoms.
- Make sure that you use the simplest combination of coefficients and double check that all # on both sides of the equation balance.
Let’s try another example:
C2H6 + O2 → CO2 + H2O
Notice that the # of carbon atoms in the reactants is 2 but there is only 1 C in the products. The # of atoms of oxygen and hydrogen are also uneven; so we have to balance the #s by adding coefficients.
First inclination is to look at the carbon atom
Add a coefficient of 2 in front of CO2 to balance the carbon atoms
Next turn to hydrogen atoms
Add coefficient of 3 in front of H2O to balance the hydrogen atoms
Now balance the remaining oxygen atoms
There is already a 2 in front of CO2 so that cannot change now. The only places where a coefficient can be added is in front of O2 and H2O. However, notice how there is no value that when placed in front of H2O and that when added to the 4 oxygen atoms in CO2, would yield an even number. Why does it have to be even? Because any value placed in front of O2 would yield an even number. Additionally, adding a coefficient in front of H2O would also disrupt the balance of H atoms.
Back to the drawing board! In the first trial, we left C2H6 alone. Let’s try a 2 in front of C2H6 and follow the same process to see if we get a better result.
Add a 4 in front of CO2 to balance carbon
Now balance the H in the products
And finally the O
Success! All the atoms are balanced. Double check each side
| Reactant Atoms | Product Atoms | |
| C | 4 | 4 |
| H | 12 | 12 |
| O | 14 | 14 |
It is important to apply the moto : “if at first you don’t succeed, right, right again” when balancing chemical equations because the first try might not work.
Also note that coefficients placed in front of a formula apply to and change the # of atoms in the entire formula, including polyatomic ions!