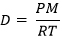The Ideal or Universal Gas Law can also be used to calculate the density or molecular mass (mass of 1 mol) of a gas. A convenient equation has been derived to make calculations a breeze (no pun intended!)
Where:
- D= Density
- P= Pressure
- R= Ideal Gas Constant (0.0821 Latm/molK)
- M= Molecular Mass of gas
- T= Temperature
HINT: You may not be given density, but be resourceful. If you have the mass and volume of the gas instead, use the density equation and you’re in business!
NOTE: Also note that since this a variation of the Ideal Gas Law, you will still be using “R.”
Problem: A 2.10-L vessel contains 4.65 g of a gas at 1.00 atm and 27.0°C. What is the molar mass of the gas?
GIVEN: